Search Healthcare Jobs
📰 FEATUREDHealthecareer Today Story
UK outlines regulatory pathway for space medicinesUK outlines regulatory pathway for medicines manufactured in space
The UK government has announced a new regulatory framework that it considers to be among the most advanced in the world for supporting the development of medicines produced in space. This initiative provides clearer regulations and guidance for companies interested in studying how microgravity conditions can influence pharmaceutical research and manufacturing processes.
Producing medicines in space presents several potential benefits. In a microgravity environment, scientists can create more precise crystal structures and stable biological materials compared to those formed on Earth — potentially contributing to more effective drugs for complicated conditions.
Through this regulatory pathway, the UK aims to strengthen cooperation between the space industry and the life sciences sector, supporting safety, quality, and regulatory standards for pharmaceutical products used on Earth.
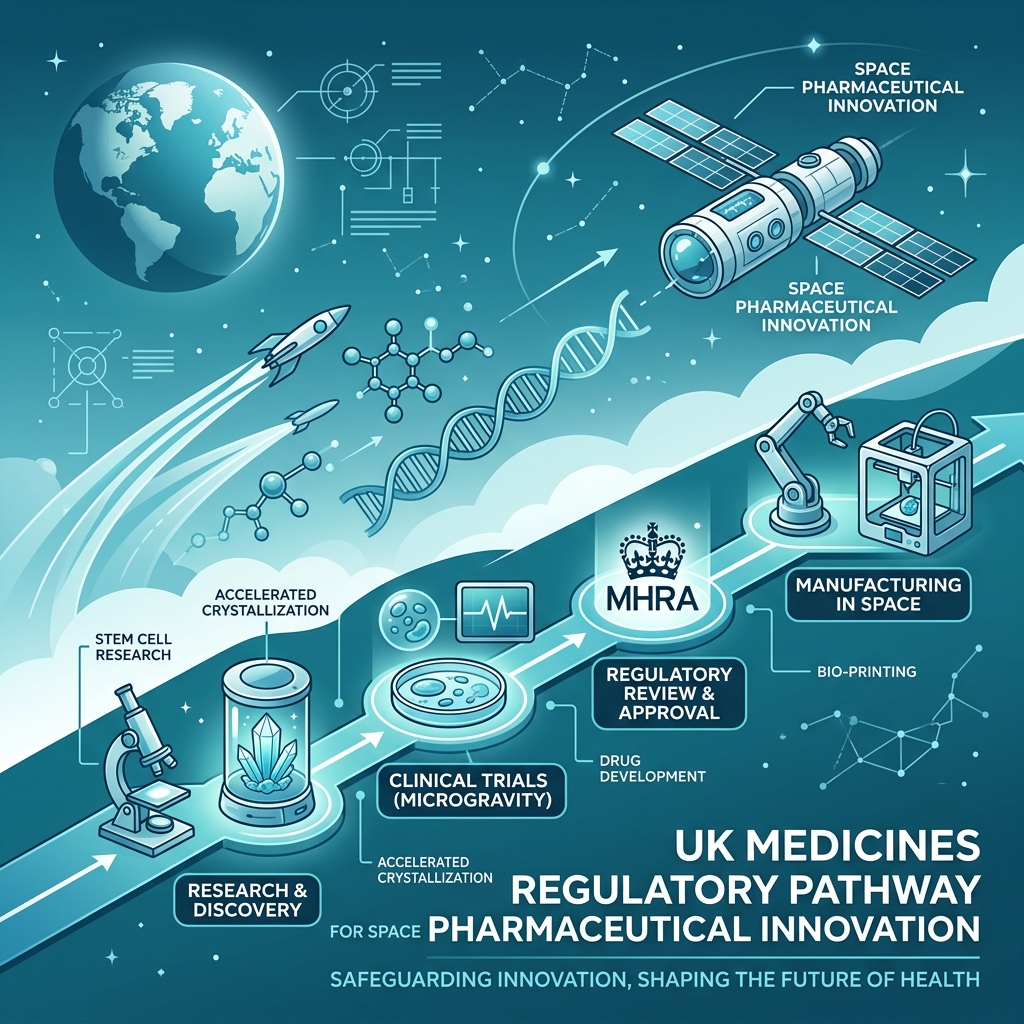
Space Pharma Innovation
UK Regulatory Framework 2026
📰 Articles
more articles » Career Advice
Career AdviceWhat are the biggest recruitment fraud risks facing you in 2026?
12-Feb-2026
 Technology⭐ Featured
Technology⭐ FeaturedAI in Clinical Trials: Use Cases, Benefits & What's Next in Healthcare Research
05-Feb-2026
 Industry
IndustryKey Clinical Research Roles Every Life Sciences Professional Should Know
27-Nov-2025
 Research
ResearchUK Biobank and Its Growing Importance in the Future of UK Life Sciences
25-Nov-2025
 Career Advice
Career AdviceThe Best Networking Strategies for Senior Professionals in Life Sciences
20-Nov-2025
 Strategy
Strategy7 Key Steps to a Successful Drug Launch Strategy in 2025
30-Sep-2025
📅 Featured Events
more events »25 Jan 2026 – 27 Jan 2026
20 Jun 2026 – 21 Jun 2026
28 Mar 2026 – 29 Mar 2026
23 May 2026 – 24 May 2026
12 Jun 2026 – 13 Jun 2026
25 Sep 2026